|
And cobalt doesn't have this favored stability, which is why it is 3d7 4s2, ripping a 4s2 electron to form 3d8, doesn't increase the stability of the subshell because you aren't making a half-full or full subshell. 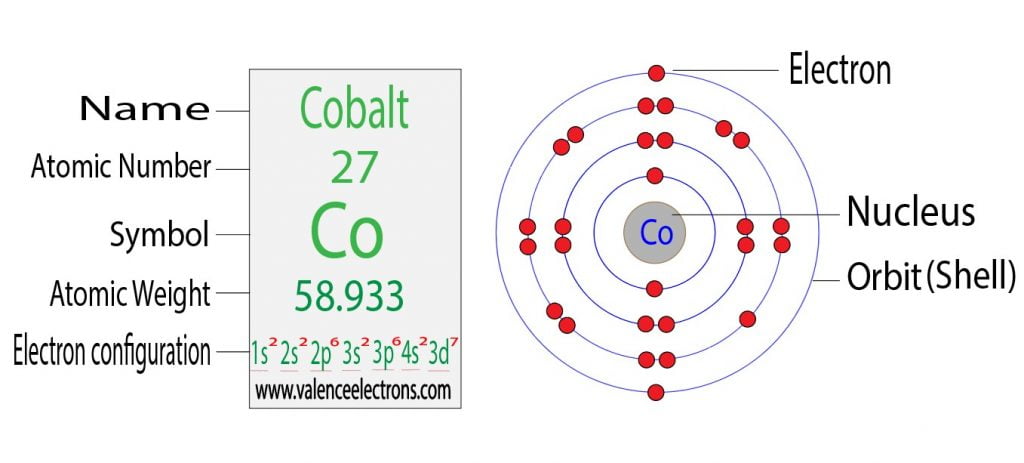
(The stability that you gain from forming a completed shell at the 3d level is more favorable than the initial energy to rip an electron away from the 4s2 shell.) This means that copper is an exception to filling of subshells, because it prefers to form a complete shell.Īnother example of an exception is Chromium, if you look up it's electron configuration it's 3d5 4s1, this is another case of when ripping one electron from the 4s2 orbital is more favorable because it forms a 3d subshell that has all of its unpaired electrons pointed the same direction (+1/2, for example).Įssentially, Chromium and Copper are exceptions in the way that their subshells are formed because of the stability of the subshell formed is higher than the initial energy required to rip that electron from the 4s2 shell. This is different from Copper, because the stability of having 3d10 is more favored than having 3d9 4s2. Someone correct me if I'm wrong, but I believe the answer to the first part of your question is that Cobalt has 5 orbitals at the 3d level, and when these orbitals are being filled, you fill one electron at a time in each one of the orbitals, so you have the first 5 electrons pointed upward (or +1/2 conformation), and then the additional 2 valence electrons form pairs with 2 of the orbitals, for a total of 7 valence electrons in the 3d7 orbital. /r/GAMSAT - Australian & UK Medical school Admission Test.P/S = Psychological, Social, and Biological Foundations of Behavior (Tentative) AAMC Sample FL Score ConversionĬ/P = Chemical and Physical Foundations of Biological SystemsĬARS = Critical Analysis and Reasoning Skillsī/B = Biological and Biochemical Foundations of Living Systems # Correct -> Scaled Score Converter for AAMC Material These posts will be removed and the user banned without warning, subject to the discretion of the mod team Learn MoreĪll of the above rules are subject to moderator discretion High Yield MCAT Links You have signed an examinee agreement, and it will be enforced on this subreddit.ĭo not intentionally advertise paid or free products or services of any sort. We have one "stickied" post for each exam and score release day, contain all test day discussion/reactions to that thread only.ĭo not discuss any specific information from your actual MCAT exam. For an example format for submitting pictures of questions from practice material click hereĭo not link to content that infringes on copyright laws (MCAT torrents, third party resources, etc).ĭo not post repeat "GOOD LUCK", "TEST SCORE", or test reaction posts. These are considered spoilers and should be marked as such. Be nice to each other, hating on other users won't help you get extra points on the MCAT, so why do it?ĭo not post any question information from any resource in the title of your post. Rudeness or trolling will not be tolerated. 
Please message the moderators with your skills/ideas! MCAT RESOURCES & INFO Study Groups Want to help us improve this subreddit or tell us about a new resource we can add to the sidebar? Below you will find our forum rules, resources, and more. 
We request that you read the sidebar COMPLETELY before you post. r/MCAT is a place for support, discussion, advice, social networking, news, study tips and more. The MCAT (Medical College Admission Test) is offered by the AAMC and is a required exam for admission to medical schools in the USA and Canada. 
All other known metals (iron, copper, silver, gold, zinc, mercury, tin, lead and bismuth) had no recorded discoverers.Welcome to the BEST place for MCAT prep and practice materials. Cobalt became the first metal to be discovered since the pre-historical period. He showed that compounds of cobalt metal were the source of the blue color in glass, which previously had been attributed to the bismuth found with cobalt. Swedish chemist Georg Brandt (1694–1768) is credited with discovering cobalt circa 1735, showing it to be a previously unknown element, distinct from bismuth and other traditional metals. Because the primary ores of cobalt always contain arsenic, smelting the ore oxidized the arsenic into the highly toxic and volatile arsenic oxide, adding to the notoriety of the ore. The first attempts to smelt those ores for copper or silver failed, yielding simply powder (cobalt(II) oxide) instead. The word cobalt is derived from the German kobalt, from kobold meaning "goblin", a superstitious term used for the ore of cobalt by miners. Discovery of cobaltCobalt has been used to color glass since the Bronze Age.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
